

IISER Chemistry - States of Matter
Exam Duration: 45 Mins Total Questions : 30
Helium atoms is two times heavier than a hydrogen molecule. At 300 K, the average K.E. of a helium atom is
- (a)
equal to that of hydrogen atom
- (b)
half that of hydrogen molecule
- (c)
same as that of hydrogen molecule
- (d)
two times that of hydrogen molecule
The vapour density of ozone would be
- (a)
24
- (b)
96
- (c)
48
- (d)
16
20 ml of nitric oxide combines with 10 ml of O2 at STP to give NO2. The final volume will be
- (a)
30 ml
- (b)
20 ml
- (c)
10 ml
- (d)
NONE OF THESE
Which one of the following gases has the greatest rate of diffusion?
- (a)
CO2
- (b)
Cl2
- (c)
NH3
- (d)
O2
A closed vessel contains equal number of nitrogen and oxygen molecules at a pressure of P. if nitrogen is removed from the system, then the pressure will be
- (a)
P
- (b)
2P
- (c)
P/2
- (d)
P2
The inversion temperature for a van der Waal's gas is given by
- (a)
\({ T }_{ i }=\frac { a }{ Rb } \)
- (b)
\({ T }_{ i }=\frac { a }{ 2Rb } \)
- (c)
\({ T }_{ i }=\frac { 2a }{ Rb } \)
- (d)
NONE OF THESE
Which of the following is a crystalline solid?
- (a)
glass
- (b)
plastic cube
- (c)
calcium oxide
- (d)
rubber ball
Frenkel defect is produced due to
- (a)
missing of one positive and one negative ion from the crystal lattice
- (b)
displacement of a cation from its proper position to an interstitial lattice site
- (c)
missing of a negative ion from its crystal lattice and the hole being occupied by an electron
- (d)
missing of a positive ion from its crystal lattice and the charge being balanced by adjacent metal ion having two charges instead of one
Which is not correct about sodium chloride crystal lattice?
- (a)
Cl- forms CCP structure and Na+ occupies octahedral holes
- (b)
Co-ordination number of chloride ion is 6
- (c)
A single unit cell has one NaCl unit in it
- (d)
Coordination number of Na+ is 6
The arrangement ABC ABC ABC ... is referred to as
- (a)
octahedral close packing
- (b)
hexagonal close packing
- (c)
tetragonal close packing
- (d)
cubic close packing
Which of the following V-t plots represents the behaviour of one mole of an ideal gas at one atmospheric pressure?
- (a)
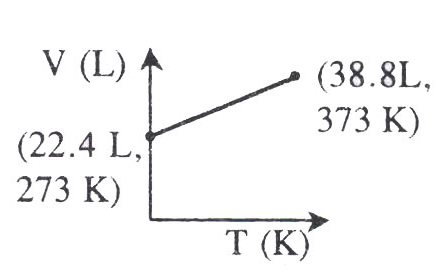
- (b)
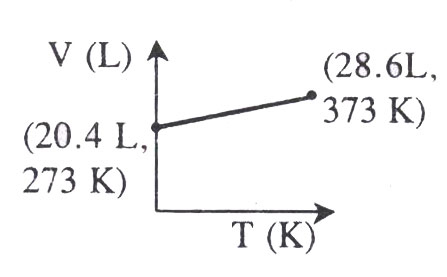
- (c)
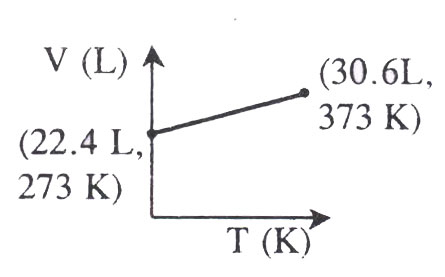
- (d)
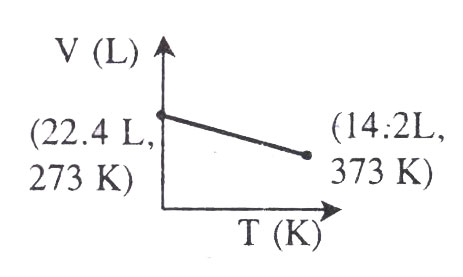
Which one of the following statements in correct?
- (a)
'He' diffuses four times faster than SO2 does
- (b)
'He' diffuses four times faster than CO2 does
- (c)
'He' diffuses four times faster than N2O does
- (d)
'He' diffuses four times faster than ClO2 does
A compound formed by elements A and B crystallises in the cubic structure, where A atoms are at the corners of a cube, while B atoms are at the face ventres. the formula of the compound would be?
- (a)
AB
- (b)
AB2
- (c)
AB3
- (d)
NONE OF THESE
In a compound Li Al(SiO3)2, the charge on SiO3 unit is
- (a)
-1
- (b)
-2
- (c)
-3
- (d)
none
The density of a gas at \({ 27 }^{ \circ }C\) and 1 atm is d. Pressure remaining constant, at which of the following temperatures will its density become 0.75 d?
- (a)
\({ 20 }^{ \circ }C\)
- (b)
\({ 30 }^{ \circ }C\)
- (c)
\(400\ K\)
- (d)
\(300\ K\)
3.2 g of oxygen (at wt. = 16) and 0.2 g of hydrogen (at wt. = 1) are placed in a 1.12 L flask at \({ 0 }^{ \circ }C\). The total pressure of the gas mixture will be
- (a)
1 atm
- (b)
4 atm
- (c)
3 atm
- (d)
2 atm
Silicon doped with arsenic is an example of which type of semiconductor?
- (a)
p-type
- (b)
n-type
- (c)
n, p type
- (d)
intrinsic
An element (atomic mass = 100 g mol-1) having BCC structure has unit cell edge 400 pm. The density of the element is
- (a)
10.370 g cm-3
- (b)
5.188 g cm-3
- (c)
7.289 g cm-3
- (d)
2.144 g cm-3
It is true that
- (a)
ZnO can act as a superconductor
- (b)
some complex metal oxides behave as superconductor at 325 K
- (c)
an impurity of tetravalent germanium in trivalent gallium creates electron deficient holes
- (d)
a Frankel defect is formed when an ion is displaced from its lattice site to an interstitial site.
Six litres of an ideal gas is at STP. The temperature of this gas was first raised to \({ 273 }^{ \circ }C\) at a constant pressure and then the pressure aws changed to 4 atm at constant temperature. The final volume of the gas would be
- (a)
2 L
- (b)
3 L
- (c)
6 L
- (d)
12 L
At \({ 0 }^{ \circ }C,\) the density of a geseous oxide at 2 bar is same as that of nitrogen at 5 bar. The molecular mass of gaseous oxide would be
- (a)
56 g mol-1
- (b)
84 g mol-1
- (c)
28 g mol-1
- (d)
70 g mol-1
If the absolute temperature of a gas having volume V cm3 is doubled and the pressure is reduced to half, the final volume would be
- (a)
2V
- (b)
4V
- (c)
8V
- (d)
16V
The ratio of most probable velocity \((\alpha)\), average velocity \((\bar { v } )\) and rootmean square velocity (u) is
- (a)
\(1:\sqrt { 2 } :\sqrt { 3 } \)
- (b)
\(\sqrt { 2 } :\sqrt { 3 } :\sqrt { 8 } \)
- (c)
\(\sqrt { 2 } :\sqrt { \frac { 8 }{ \pi } } :\sqrt { 3 } \)
- (d)
\(1:\sqrt { 8\pi } :\sqrt { 3 } \)
The drain cleaner, Drainex contains small bits of aluminium which react with caustic soda to produce hydrogen. When 0.15 g of aluminium reacts, the approximate volume of hydrogen that will be released at \({ 20 }^{ \circ }C\)
- (a)
200 ml
- (b)
400 ml
- (c)
100 ml
- (d)
500 ml
A student forgot to add the reaction mixture to the round bottomed flask at \({ 27 }^{ \circ }C\) but put it on the flame. After a lapse of time, he realised his mistake; using a pyrometer he found the temperature of the flask was \({ 477 }^{ \circ }C.\) The fraction of air expelled would be
- (a)
\(1\over 4\)
- (b)
\(2\over 3\)
- (c)
\(1\over 3\)
- (d)
\(3\over 5\)
Density of a gas is found to be 5.40 g dm-3 at \({ 27 }^{ \circ }C\) at 2 bar pressure. Its density at STP would be
- (a)
2.4 g dm-3
- (b)
3 g dm-3
- (c)
2 g dm-3
- (d)
NONE OF THESE
The rms velocity of hydrogen is \(\sqrt { 7 } \) times the rms velocity of nitrogen. If T is the temperature of the gas
- (a)
T(H2)=T(N2)
- (b)
T(H2)>T(N2)
- (c)
T(H2)<T(N2)
- (d)
\(T({ H }_{ 2 })=\sqrt { 7 } \quad T({ N }_{ 2 })\)
The value of universal gas constant R depends on
- (a)
temperature of gas
- (b)
volume of gas
- (c)
number of moles of gas
- (d)
units of volume and pressure
Value of gas constant R is
- (a)
0.082 litre atm
- (b)
0.987 cal. mol-1 K-1
- (c)
8.314 J mol-1 K-1
- (d)
83 erg mol-1 K-1
For an ideal gas, number of moles per litre in terms of its pressure P, gas constant R and temperature T is
- (a)
\(PT\over R\)
- (b)
PRT
- (c)
\(P\over RT\)
- (d)
\(RT\over P\)


1654060945.png)
1654060942.png)
1653987551.png)
1653987086.png)

1582095229.jpg)