

NEET 2020 - Chemistry Study Materials
Exam Duration: 120 Mins Total Questions : 100
Given below are few statements. Mark the statement which is not correct.
- (a)
Atoms are neighter created nor destroyed in a chemical reaction
- (b)
Law of definite proportion states that a given compound always contains exactly the same proportion of elements by weight
- (c)
G
- (d)
A pure compound has always a fixed proportion of masses of its constituents.
Which of the following formulae is not correctly depicted?
- (a)
Molar mass = \(\frac { Mass\ of\ substance }{ moles\ of\ substance } \)
- (b)
Mass of one molecule of a substance = \(\frac { gram\ molecular\ mass\ of\ the\ substance }{ Avogadr'o\ number } \)
- (c)
Number of molecules = \(\frac { Mass\ of\ the\ substance }{ Molar\ mass }\) x Agvogasro's no
- (d)
Number of moles x molar mass = number of molecules
Molar mass = mass per mole of the substance = \(\frac{Mass \space of \space substance}{Moles \space of \space substance}\)
Rewriting it as: Number of moles of a substance =\(\frac{Mass \space of \space substance}{Molar \space mass}\)
Also, number of moles of a substance \(\times\) Molar mass = mass of the substance.
Number of molecules = Number of moles \(\times\) Avogadro's constant = \(\frac{Mass \space of \space substance}{Molar \space mass}\)\(\times\) Avogadro's no.
Assertion: The mass of a substance is constant whereas its weight may vary from one place to another.
Reason : Mass of a substance is the amount of matter present in it while weight is the force exerted by gravity on an object.
- (a)
Both Assertion and Reason are correct and Reason is the correct explanation for Assertion
- (b)
Both Assertion and Reason are correct but Reason is not the correct explanation for Assertion
- (c)
Assertion is correct but Reason is incorrect
- (d)
Both Assertion and Reason are incorrect
The weight of a substance may vary from one place to another due to change in gravity but mass of any substance remains the same.
Effective nuclear charge (Zeff) for a nucleus of an atom is defined as
- (a)
shielding of the outermost shell electrons from the nucleus by the innermost shell electrons
- (b)
the net positive charge experienced by electron from the nucleus
- (c)
the attractive force experienced by the nucleus from electron
- (d)
screening of positive charge on nucleus by innermost shell electrons
The effective nuclear charge Zeff is the net positive charge experienced by an electron from the nucleus in a polyelectronic atom.
The term "effective" is used because the shielding effect of negatively charged electrons prevents higher orbital electrons from experiencing the full nuclear charge of the nucleus due to the repelling effect of inner-layer electrons.
What is the electronic configuration of O2- ion?
- (a)
1s2 2s2 2p6
- (b)
1s2 2s2 2p4
- (c)
1s2 2s2 2p5
- (d)
1s2 2s2 2p3
O2-:
The atomic number of O = 8 = number of electrons
Number of electrons in O2- are 8 + 2 = 10
Electronic configuration will be 1s22s22p6
The velocity of an electron in a certain Bohr orbit of H-atom bears the ratio 1:275 to the velocity of light. The quantum number (n) of the orbit is
- (a)
3
- (b)
2
- (c)
1
- (d)
4
Velocity of electrons =\(\frac { 1 }{ 275 } \) x velocity of light
=\(\frac { 1 }{ 275 } \) x 3 x 1010 = 1.09 x 108 cm s-1
Since vn=\(\frac { 2\pi { e }^{ 2 } }{ nh } \)
∴ 1.09 x 108 = \(\frac { 2\times 3.14\times (4.803\times 10^{ -10 })^{ 2 } }{ 6.626\times 10^{ -27 }\times n } \)
∴ n=2
What is the name and symbol of the element with atomic number 112?
- (a)
Ununbium, Uub
- (b)
Unnilbium, Unb
- (c)
Ununnillum, Uun
- (d)
Ununtrium, Uut
The name and symbol of the element with atomic number 112 are Ununbium and Uub respectively. Alternatively, the name and symbol of the element with atomic number 112 are Copernicium and Cn respectively.
An element with atomic number 117 is known as
- (a)
nihonium
- (b)
flerovium
- (c)
tennessine
- (d)
roentgenium
An element with atomic number 117 is known as tennessine. It is also called Ununseptium and has symbol Uus. Roentgenium has atomic numbe 111 and symbol Uuu/Rg.
Which of the following elements shown as pairs with their atomic numbers belong to the same period?
- (a)
Z = 19 and Z = 38
- (b)
Z = 12 and Z = 17
- (c)
Z = 11 and Z = 21
- (d)
Z = 16 and Z = 35
Same period elements have same outer most shell (n)
Z = 19 is [Ar]4s1 and Z=38 is [Kr]5s2: different n=4 and 5
Z = 12 is [Ne]3s2 and Z=17 is [Ne]3s23p5: smae n=3
Z = 11 is [Ne]3s1 and Z=21 is [Ar]4s23d1: different n = 3 and 4
Z = 16 is [Ne]3s23p4 and Z = 35 is [Ar]4s24p5: different n = 3 and 4
Mark the incorrect statement in the following :
- (a)
The bond order in the species O2, O2+ and O2- decreases as O2+ > O2 > O2-
- (b)
The bond energy in a diatomic molecule always increases when an electron is lost
- (c)
Electrons in antibonding MO contribute to repulsion between two atoms
- (d)
With increase in bond order, bond length decrease and bond strength increases
It is not necessary that removal of an electron increases the bond energy. However, it depends on the orbital from which electron is removed. If electron is removed from a bonding orbital then the bond order decreases, bond length increases and bond energy decreases. If electron is removed from antibonding orbital then bond order increases, bond length decreases and energy increases.
Two elements P and Q combine to form a compound. If P has 2 and Q has 6 electrons in their outermost shell, what will be formula of the compound formed?
- (a)
PQ
- (b)
P2Q
- (c)
P2Q3
- (d)
PQ2
Valence electrons in Pis 2; Valence electrons in Q is 6.
P- 2e-\(\rightarrow \) p2+
Q+2e- \(\rightarrow \) Q2-
Since both show a valency of 2, the formula for the compound will be PQ.
Hydrogen bonds are formed in many compounds e.g., H2O, HF, NH3. The boiling point of such compounds depends to a large extent on the strength of hydrogen bond and the number of hydrogen bonds. The correct decreasing order of the boiling points of above compounds is
- (a)
\(HF>H_{ 2 }O>NH_{ 3 }\)
- (b)
\(H_{ 2 }O>HF>NH_{ 3 }\ \)
- (c)
\(HF>H_{ 2 }O>NH_{ 3 }\)
- (d)
\(NH_{ 3 }>H_{ 2 }O>HF\)
Strength of hydrogen bonding depends on the size and electronegativity of the atom. Smaller the size of the atom, greater is the electronegativity and hence stronger is the H-bonding. Thus, the order of strength of H-bonding is H ...F > H ...O > H ...N. But each HF molecule is linked only to two other HF molecules while each H2O molecule is linked to four other H2O molecules through H-bonding.
Hence, the decreasing order of boiling points is \(H_{ 2 }O>HF>NH_{ 3 }\ \)
Two gases A and B having the same volume diffuse through a porous partition in 20 and 10 s respectively. The molecular mass of A is 49 U. Molecular mass of B will be :
- (a)
12.25u
- (b)
6.50u
- (c)
25.00u
- (d)
50.00u
According to Graham's diffusion law
\(\frac { { r }_{ A } }{ { r }_{ B } } =\sqrt { \frac { { M }_{ B } }{ { M }_{ B } } } \)⇒\(\frac { { V_{ A } }/{ { t }_{ A } } }{ { { V }_{ B } }/{ { t }_{ B } } } =\sqrt { \frac { { M }_{ B } }{ { M }_{ A } } } \)
Given: VA = VB
\(\frac { { V }/{ 20s } }{ { V }/{ 10s } } =\sqrt { \frac { { M }_{ B } }{ 49 } } \)⇒\(\frac { 1 }{ 2 } =\sqrt { \frac { { M }_{ B } }{ 49 } } \)
MB=\(\frac { 1 }{ 4 } \)x 49=12.25 u
At NTP the volume of a gas is 40 mL. If pressure is increased to 800 mm of Hg at the same temperature, what will be the volume of the gas?
- (a)
38 mL
- (b)
22400 mL
- (c)
240 mL
- (d)
431 mL
P1 V1 = P2 V2 or 760 x 40 = 800 X V2
\({ V }_{ 2 }=\frac { 760\times 40 }{ 800 } =38mL\)
For the reactions, N2 + 3H2 ⟶ 2NH3, ∆H = ?
- (a)
∆E + 2RT
- (b)
∆E - 2RT
- (c)
∆H = RT
- (d)
∆E - RT
For the given reaction,
N2(g) + 3H2(g) ⟶ 2NH3(g)
∆ng = 2 - (1 + 3) = -2
Now, ∆H = ∆E + ∆ng RT
∆H = ∆E- 2 RT
If ∆H is the change in enthalpy and ∆E, the change in internal energy accompanying a gaseous reaction?
- (a)
∆H is always greater than ∆E
- (b)
∆H < ∆E only if the number of moles of products is greater than the number of moles of the reactants
- (c)
∆H is always less than ∆E
- (d)
∆H < ∆E only if the number of moles of products is less than the number of moles of the reactants
We know that
\(\triangle\)H = \(\triangle\)E + \(\triangle\)(PV)
\(\triangle\)H = \(\triangle\)E + \(\triangle\)(nRT)
When number of moles in product is less than number of moles in reactant.
So, \(\triangle\)n = -ve
So, \(\triangle\)H < \(\triangle\)E
PCl5 , PCl3 and Cl2 are at equilibrium at 500 K with concentration 2.1 M PCl3, 2.1 M Cl2 and 1.9 M PCl5 , The equilibrium constant for the given reaction is
PCl5(g) \(\rightleftharpoons \) PCl 3(g) + Cl2(g)
- (a)
2.32
- (b)
1.79
- (c)
4.2
- (d)
3.8
PCl5 ⇌ PCl3 + Cl2
\(K_{c}=\frac{[PCl_{3}][Cl_{2}]}{[PCl_{5}]}\)
\(K_{c}=\frac{2.1\times2.1}{1.9}\)
\(K_{c}=\frac{4.41}{1.9}\)
\(\therefore\) the equilibrium constant of the above reaction Kc = 2.321
On electrolysis of dilute sulphuric acid using platinum electrodes, the product obtained at the anode will be :
- (a)
Hydrogen
- (b)
Oxygen
- (c)
Hydrogen Sulphide
- (d)
Sulphur Dioxide
Product obtained at anode will be oxygen. During electrolysis:
At cathode: Reduction reaction occurs.
2H+ (aq) + 2e- ⟶ H2 (g) (hydrogen gas)
At anode: Oxidation reaction occurs.
2H2O (l) ⟶ 4H+ (aq) + O2 (g) + 4e- (oxygen gas)
What will be the balanced equation in acidic medium for the given reaction?
\(Cr_{2}O^{2-}_{7(aq)}+SO_{2(g)}\rightarrow Cr^{3+}_{(aq)}+SO^{2-}_{4(aq)} \)
- (a)
\(Cr_{2}O^{2-}_{7(aq)}+3SO_{2(g)}+ 2H^{+}_{(aq)}\rightarrow 2Cr^{3+}_{(aq)}+3SO^{2-}_{4(aq)}+H_{2}O_{(l)} \)
- (b)
\(2Cr_{2}O^{2-}_{7(aq)}+3SO_{2(g)} +4H^{+}_{(aq)}\rightarrow 4Cr^{3+}_{(aq)}+3SO^{2-}_{4(aq)}+2H_{2}O_{(l)} \)
- (c)
\(Cr_{2}O^{2-}_{7(aq)}+3SO_{2(g)} +14H^{+}_{(aq)}\rightarrow 2Cr^{3+}_{(aq)}+3SO^{2-}_{4(aq)}+7H_{2}O_{(l)} \)
- (d)
\(Cr_{2}O^{2-}_{7(aq)}+6SO_{2(g)}+ 7H^{+}_{(aq)}\rightarrow 2Cr^{3+}_{(aq)}+6SO^{2-}_{4(aq)}+7H_{2}O_{(l)} \)
\(Cr_{2}O^{2-}_{7(aq)}+SO_{2} \rightarrow 2Cr^{3+}+6SO^{2-}_{4}\) (in acidic solution) Oxidation half equation:
\(SO_{2}+2H_{2}O \rightarrow SO^{2-}_{4}+4H^{+}+2e^{-}\) -- (i)
Reduction half equation:
\(Cr_{2}O^{2-}_{7(aq)}+14H^{+}+6e^{-}\rightarrow 2Cr^{3+}+7H_{2}O\) --- (ii)
Multiplying eqn. (i) by 3 and adding to eqn. (ii) we get
\(Cr_{2}O^{2-}_{7(aq)}+3SO_{2(g)}+ 2H^{+}_{(aq)}\rightarrow 2Cr^{3+}_{(aq)}+3SO^{2-}_{4(aq)}+H_{2}O_{(l)} \)
In the following question, a statement of assertion is followed by a statement of reason. Mark the correct choice as :
Assertion: A metal having negative reduction potential when dipped in the solution of its own ions has a tendency to pass into solution.
Reason: Metals undergo reduction.
- (a)
If both assertion and reason are true and reason is the correct explanation of assertion.
- (b)
If both assertion and reason are true but reason is not the correct explanation of assertion
- (c)
If assertion is true but reason is false.
- (d)
If both assertion and reason are false.
A metal having negative reduction potential has high tendency to get oxidised and hence metal passes into the solution.
Non-stoichiometric hydrides are produced by
- (a)
palladium, vanadium
- (b)
manganese, lithium
- (c)
nitrogen, fluorine
- (d)
carbon, nickel
The hydrogen deficient compounds formed by the reaction of d−block and f−block elements with dihydrogen are called Non-stoichiometric compounds.
The d-block and f-block element form non-stoichiometric hydride because of the vacant d- and f-orbitals along with the small size.
Their elemental composition proportions cannot be represented in integers. They disobey the law of constant composition. Among the elements given, only vanadium and palladium form non-stoichiometric hydrides.
The formula for permutit or zeolite which is used as softner in ion-exchange method is
- (a)
NaAISiO4
- (b)
NaAIO2
- (c)
Ca3(PO4)2
- (d)
Na2SO4
Permutit is an artificial zeolite. It is sodium aluminium orthosilicate
Na2Al2Si2O8.xH2O.
Na2Al2Si2O8.xH2O+Ca2+(orMg2+)→CaAl2Si2O8.xH2O+2Na+
permutit exhausted permutit
Match List-I with List-II. Choose the correct matching codes from the choices given
| List I (Hydride) |
List II (Type of Hydride) |
||
|---|---|---|---|
| A. | BeH2 | 1. | Complex |
| B. | AsH3 | 2. | Lewis acid |
| C. | B2H6 | 3. | Interstitial |
| D. | LaH3 | 4. | Covalent |
| E | LiAIH4 | 5. | Intermediate |
- (a)
A-6, B-2, C-4, D-5, E-l
- (b)
A-6, B-2, C-4, D-3, E-l
- (c)
A-6, B-4, C-2, D-3, E-5
- (d)
A-5, B-4, C-2, D-3, E-l
Complex: Addition compounds which do not give all their constituent ions when dissolved in water. Here, the individual properties of ions are lost. e.g., [Cu(NH3)4]SO4, LiAIH4
Lewis acid: Electron deficient species which gain electrons while forming a bond with Lewis bases.
\({ B }_{ 2 }{ H }_{ 6 }\xrightarrow { low\ temp }{ B }_{ 2 }{ H }_{ 6 }.2{ NH }_{ 3 }\) (adduct)
Interstitial metallic hydrides: Many f-block hydrides are non-stoichiometric, e.g., LaHn, TiHn, etc. where the chemical composition is variable e.g., LaH2.87, YbH2.5, etc. Such compounds are called interstitial compounds. BeH2: Intermediate hydride and polymeric in nature. AsH3: Covalent.
Which one of the following properties of alkali metals increases in magnitude as the atomic number rises?
- (a)
Ionic radius
- (b)
Melting point
- (c)
Electronegativity
- (d)
First ionization energy
The ionic radii of alkali metal increases as the atomic number increases. Because on moving top to bottom in the group, increase in the number of shell's takes place and hence, ionic radii increases.
When sodium reacts with excess of oxygen, the oxidation number of oxygen changes from:
- (a)
0 to -1
- (b)
0 to -2
- (c)
-1 to -2
- (d)
No change
when sodium reacts with excess of oxygen, it lead to the formation of metal oxides such as sodium peroxide.
0 -1
2Na + O2 → Na2O2
hence there is a change in oxidation state from 0 to -1.
The properties of Li are similar to those of Mg. This is because:
- (a)
both have nearly the same size.
- (b)
both has their charge to size ratio nearly the same.
- (c)
both have similar electronic configurations
- (d)
both are found together in nature
The properties of lithium are similar to those of Mg because they are having diagonal relationship.
The elements having diagonal relationship show similar properties and the reason is because the both of them will have similar electronegativity and polarising power i.e. charge per size ratio of both the elements are nearly the same.
The formula of soda ash is
- (a)
Na2CO3.10H2O
- (b)
Na2CO3.2H2O
- (c)
Na2CO3.H2O
- (d)
Na2CO3
Anhydrous Na2 CO3 is called soda ash while sodium carbonate decahydrate, Na2CO3. 10H2O is called washing soda
Reaction of HBr with propene in the presence of peroxide gives:
- (a)
iso - propyl bromide
- (b)
3 - bromo propane
- (c)
allyl bromide
- (d)
n - propyl bromide
Reaction of HBr with propene in the presence of peroxide gives n-propyl bromide. This addition reaction is an example of anti - Markovnikov's addition reaction.
(i.e., it is completed in form of free radical addition)
CH3 - CH = CH2 + HBr \(\underrightarrow { peroxide } \) \({ CH }_{ 3 }-\underset { n-propylbromide }{ { CH }_{ 2 } } -{ CH }_{ 2 }Br\)
Which of the following reactions is taking place resulting in discolouration of marble of the buildings like Taj Mahal?
- (a)
CaCO3 + H2SO4 \(\rightarrow\) CaSO4 + H2O + CO2
- (b)
CaCO3 + 2HCI \(\rightarrow\) CaCl2 + H2O + CO2
- (c)
CaCO3 + H2O \(\rightarrow\) Ca(OH)2 + CO2
- (d)
CaCO3 \(\rightarrow\) CaO + CO2
Acid rain is the state when the pH of rain falls below 5.6. Acid rain damages the buildings and statues as they dissolve and corrodes the heavy metals. This happens when sulphuric acid nitric acids in the polluted air react with calcite in marble, the calcite dissolves. It also dissolves metals from soils, rocks and sediments.
Freons are not recommended to be used in refrigerators because they:
- (a)
cause global warming
- (b)
cause acid rain
- (c)
cause depletion of ozone layer
- (d)
cause very less cooling
Freon is basically a gas which is used for cooling. It is non-flammable substance, but if it leaks it smells like fresh-cut grass. As the CFC's causes depletion of the ozone layer, freons are not used as coolants anymore.
Match the column I with column II and mark the appropriate choice.
| Column I | Column II | ||
| (A) | Peroxyacetyl nitrate | (i) | Global warming |
| (B) | Polychlorinated biphenyls | (ii) | Photochemical smog |
| (C) | Dioxides of carbon and sulphur | (iii) | Water pollutant |
| (D) | IR active molecules | (iv) | Acid rain |
- (a)
(A) \(\rightarrow\) (ii), (B) \(\rightarrow\) (iii), C \(\rightarrow\) (iv), (D) \(\rightarrow\) (i)
- (b)
(A) \(\rightarrow\) (iii), (B) \(\rightarrow\) (iv), C \(\rightarrow\) (ii), (D) \(\rightarrow\) (i)
- (c)
(A) \(\rightarrow\) (iv), (B) \(\rightarrow\) (ii), C \(\rightarrow\) (iii), (D) \(\rightarrow\) (i)
- (d)
(A) \(\rightarrow\) (i), (B) \(\rightarrow\) (iii), C \(\rightarrow\) (ii), (D) \(\rightarrow\) (iv)
Peroxyacetyl nitrate is a secondary pollutant since they form in the atmosphere after the emission of primary pollutants. It is present in photochemical smog.
Surface water getting contaminated with PCB waste generally has high levels of PCBs in sediment, as the PCBs attach to organic matter. PCBs can be slowly released from the sediment into the water and evaporate into the air.
Sulfur dioxide another toxic chemical gas when combines with water and air, forms sulfuric acid, which is the main component of acid rain. Carbon dioxide in the air can dissolve in rainwater to form carbonic acid, H2CO3 and contributes to acid rain.
IR active molecules in the atmosphere can increase their vibrational energy by absorbing some of this infrared light, and thus increasing the temperature of the atmosphere resulting in Global warming.
Solid X is a very hard solid which is electrical insulator in solid as well as in molten state and has extremely high melting point. What type of solid is it?
- (a)
Ionic solid
- (b)
Covalent solid
- (c)
Metallic solid
- (d)
Molecular solid
Covalent solids have high melting point due to their interconnected covalent bonds. Since there are no free electrons present in covalent solids, so they do not conduct electricity in solid state as well as molten state. Some of the examples are Diamond and Quartz.
Alkali halides do not show Frenkel defect because:
- (a)
cations and anions have almost equal size
- (b)
there is a large difference in size of cations and anions
- (c)
cations and anions have low coordination number
- (d)
anions cannot be accommodated in voids
Frenkel defect is possible in the compounds in which the size of cations and anions are different. In this defect, the smaller ion comes to interstitial place.
In Alkali halides the size of cations and anions is almost equal. So the ion size is large and they cannot get into the interstitial site.
So, these kinds of compounds do not show Frenkel defect.
Experimentally it was found that a metal oxide has formula M0.98O. Metal M, is present as M2+ and M3+ in its oxide. Fraction of the metal which exists as M3+ would be:
- (a)
5.08%
- (b)
7.01%
- (c)
4.08%
- (d)
6.05%
Since the oxidation state of oxygen is −2. So, for M0.980 to be neutral, the total oxidation state of M0.98 has to be +2.
Let the fraction of M3+ be x.
Then fraction of M2+ will be (0.98−x).
Now for the compound to be neutal,
3x + 2(0.98 − x) = 2
3x + 1.96 − 2x = 2
x = 2 - 1.96
x = .04
So, fraction of M3+ will be 4%.
Assertion: Packing efficiency of body centred cubic structure is 68%.
Reason : 68% is the maximum packing efficiency any crystal can have
- (a)
If both assertion and reason are true and reason is the correct explanation of assertion.
- (b)
If both assertion and reason are true but reason is not the correct explanation of assertion
- (c)
If assertion is true but reason is false.
- (d)
If both assertion and reason are false
Packing efficiency of various crystals is given as:
Face-centered cube - 74%
Body-centered cube - 68%
Simple Cube - 52.4%
In the body-centered cubic unit cell, the atoms are present at corners and body center of the cube. So, the effective number of atoms in unit cell =2
If the radius of the atom is r and edge length of the cube is a In body-centered cubic unit cell atoms along the body, diagonals touch each other.
So, \(\sqrt{3}a=4r\)
\(\Rightarrow r=\frac{\sqrt{3}}{4}a\)
So, volume occupied \(=2 \times \frac{4}{3}\times\pi r^{3}\)|
Total volume =a3 =\(\left(\frac{4}{\sqrt{3}^{r}}\right)^{3}\)
So, packing efficiency = \(\frac{2\times\frac{4}{3}\pi r^{3}}{\frac{4}{\sqrt{3}}^{3}r^{3}}\times100%\)%
\(=\frac{\pi\times2\sqrt{3}}{16}\times\)100%=68%
Which one of the following electrolytes has the same value of van't Hoff factor (i) as that of Al2(SO4)3 if all are 100% ionized?
- (a)
K2SO4
- (b)
K3[Fe(CN)6]
- (c)
Al(NO3)
- (d)
K4[Fe(CN)6]
For Al2(SO2)3
Al2(SO4)3 ⇌ 2Al+3 + 3SO42-
van't Hoff factor, i = 5
For K2SO4,
K2SO4 ⇌ 2K+ + SO42- (i = 3)
For K3[Fe(CN)6],
K3[Fe(CN)6] ⇌ 3K+ + [Fe(CN)6]2-(i = 4)
For Al(NO3)3,
Al(NO3)3 ⇌ Al3+ + 3NO3- (i = 4)
For K4[Fe(N)6]
K4[Fe(CN)6]⇌4K+ + [Fe(CN)6]4-(i = 5)
Sprinkling of salt helps in clearing the snow covered roads in hills. The phenomenon involved in the process is
- (a)
lowering in vapour pressure of snow
- (b)
depression in freezing point of snow
- (c)
increase in freezing point of snow
- (d)
melting of ice due to increase in temperature by putting salt.
When salt is spread over snow, snow starts melting from the surface because of depression in freezing point and helps in clearing the roads.
What will be the osmotic pressure in pascals exerted by a solution prepared by dissolving 1.0g of polymer of molar mass 150,000 in 500 mL of water at 37°C?
- (a)
30.96 Pa
- (b)
34.36 Pa
- (c)
68.72 Pa
- (d)
48.25 Pa
No. of moles of polymer =\(\frac{1}{150,000}\)
\(\pi =CRT=\frac { n }{ V } RT\)
\(\pi =\frac { 1 }{ 150,000 } \times \frac { 8.314\times { 10 }^{ 3 }\times 310 }{ 0.5 } \)=34.36 pa
(R = 8.314 x 103 Pa L K-1 mol-1)
On the basis of information given below mark the correct option.
Information: On adding acetone to methanol some of the hydrogen bonds between methanol molecules break.
- (a)
At specific composition methanol-acetone mixture will form minimum boiling azeotrope and will show positive deviation from Raoult's law.
- (b)
At specific composition methanol-acetone mixture forms maximum boiling azeotrope and will show positive deviation from Raoult's law.
- (c)
At specific composition methanol-acetone mixture will form minimum boiling azeotrope and will show negative deviation from Raoults law.
- (d)
At specific composition methanol-acetone mixture will form maximum boiling azeotrope and will show negative deviation from Raoult's law.
Assertion: Pressure does not have any effect on solubility of solids in liquids.
Reason: Solids and liquids are highly incompressible
- (a)
If both assertion and reason are true and reason is the correct explanation of assertion.
- (b)
If both assertion and reason are true but reason is not the correct explanation of assertion.
- (c)
If assertion is true but reason is false.
- (d)
If both assertion and reason are false
The equivalent conductance of Ba2+ and Cl- are 127 and 76 Ω-l cm-1 eq-1 respectively at infinite dilution. The equivalent conductance of BaCl2 at infinite dilution will be:
- (a)
139.52
- (b)
203
- (c)
279
- (d)
101.5
According to Kohlrausch's law, the equivalent conductance of BaCl2 at infinite dilution,
λ∞ of BaCl2 = \(\frac { 1 }{ 2 } { \lambda }_{ \infty }\) of Ba2+ + λ∞ of Cl-
\(\frac { 127 }{ 2 } +76={ \lambda }_{ \infty }\) = 139.5
Standard electron potential of three metals X, Y and Z are -1.2 V + 0.5 V and -3.0 V respectively. The reducing power of these metals will be :
- (a)
Y > X > Z
- (b)
Z > X > Y
- (c)
X > Y > Z
- (d)
Y > Z > X
E0x= -1.2 V, E0y = 0.5 V, E0z= -3.0 V
∴ Z > X > Y (As higher the reduction potential, lesser the reducing power)
Assertion: The conductivity of electrolytic solutions increases with increase of temperature.
Reason : Electronic conductance decreases with increase of temperature.
- (a)
If both assertion and reason are true and reason is the correct explanation of assertion.
- (b)
If both assertion and reason are true but reason is not the correct explanation of assertion.
- (c)
If assertion is true but reason is false.
- (d)
If both assertion and reason are false.
Conductivity of electrolytic solutions depends upon the ions produced in solution which increases with increase of temperature. (ionization increases).
Electrical conductance through metals is called metallic or electronic conductance and is due to the movement of electrons. The electronic conductance depends on
- (a)
the nature and structure of the metal
- (b)
the number of valence electrons per atom
- (c)
change in temperature
- (d)
all of these.
Specific conductance of 0.1 M NaCl solution is 1.01 x 10-2 ohm-1 cm-1 Its molar conductance in ohm-1 cm-2 mol-1 is
- (a)
1.01 x 102
- (b)
1.01 x 103
- (c)
1.01 x 104
- (d)
1.01
\({ A }_{ m }=\frac { k\times 1000 }{ M } \)
\(\frac { 1.01\times { 10 }^{ -2 }\times 1000 }{ 0.1 } \) = 1.01 x 102 ohm-1 cm2 mol-1
How many coulombs of electricity is required to reduce 1 mole of \({ Cr }_{ 2 }{ O }_{ 7 }^{ 2- }\)in acidic medium?
- (a)
4 x 96500 C
- (b)
6 x 96500 C
- (c)
2 x 96500 C
- (d)
1 x 96500 C
In acidic medium, dichromate ions are reduced according to the equation,
\({ Cr }_{ 2 }{ O }_{ 7 }^{ 2- }+14{ H }^{ - }+6{ e }^{ - }\rightarrow { 2Cr }^{ 3+ }+{ 7H }_{ 2 }O\)
1 mole of \({ Cr }_{ 2 }{ O }_{ 7 }^{ 2- }\) ion requires 6 moles of electrons or 6 x 96500 C of electricity.
Assertion: For the reaction CHCl3 + Cl2 ⟶ CCl4 + HCl
Rate =k[CHCl3][Cl2]1/2
Reason: Rate of reaction is always equal to the sum of the stoichiometric coefficients of the reacting species in a balanced chemical equation.
- (a)
If both assertion and reason are true and reason is the correct explanation of assertion
- (b)
If both assertion and reason are true but reason is not the correct explanation of assertion
- (c)
If assertion is true but reason is false
- (d)
If both assertion and reason are false
Rate of reaction depends upon the experimental conditions such as concentration of reactants, temperature and catalyst. It mayor may not be equal to the stoichiometric coefficients of the reacting species in a balanced chemical equation.
The rate constant for the reaction, 2N2O5 ⟶ 4NO2 + O2 is 2 x 10-5 s-1. If rate of reaction is 1.4 x 10-5 mol L-1 s-1, what will be the concentration of N2O5 in mol L-1?
- (a)
0.8
- (b)
0.7
- (c)
1.2
- (d)
1
Rate = k[N2O5] (first order as unit of rate constant is s-1.
[N2O5]=\(\frac { rate }{ k } =\frac { 1.4\times { 10 }^{ -5 }mol{ L }^{ -1 }{ s }^{ -1 } }{ 2\times { 10 }^{ -5 }{ s }^{ -1 } } \)=0.7 mol L-1 .
The activity of an enzyme becomes ineffective
- (a)
at low temperature
- (b)
at atmospheric pressure
- (c)
at high temperature
- (d)
in aqueous medium
Enzymes are effective only at optimum temperature. They become ineffective at very high temperature.
A lyophobic colloid cannot be formed by
- (a)
mixing dispersed phase and dispersion medium
- (b)
chemical reactions like hydrolysis
- (c)
exchange of solvent
- (d)
peptisation
Which of the following is not an explanation for the origin of charge on the colloidal particles?
- (a)
Due to frictional electrification
- (b)
Due to dissociation of surface molecules
- (c)
Due to electrophoresis
- (d)
Due to selective adsorption of ions
Due to the presence of charge on colloidal particles, electrophoresis takes place.
Assertion: Hydrolysis of ester is an example of auto-catalytic reaction.
Reason: A catalyst speeds up the process without participating in the mechanism.
- (a)
If both assertion and reason are true and reason is the correct explanation of assertion.
- (b)
If both assertion and reason are true but reason is not the correct explanation of assertion.
- (c)
If assertion is true but reason is false.
- (d)
If both assertion and reason are false.
A catalyst participates in the mechanism of the reaction and is regenerated after the reaction.
Assertion: A colloidal sol scatters light but a true solution does not.
Reason: The particles in a colloidal sol move slowly than in a true solution.
- (a)
If both assertion and reason are true and reason is the correct explanation of assertion.
- (b)
If both assertion and reason are true but reason is not the correct explanation of assertion.
- (c)
If assertion is true but reason is false.
- (d)
If both assertion and reason are false.
How do we separate two sulphide ores by froth floatation method?
- (a)
By using excess of pine oil
- (b)
By adjusting proportion of oil to water or using depressant
- (c)
By using collectors and froth stabilisers like xanthates.
- (d)
By using some solvent in which one of the sulphides is soluble.
By using depressants or adjusting proportion of oil to water, selective prevention of one sulphide ore from coming to the froth is possible.
In blast furnace iron oxide is reduced by
- (a)
silica
- (b)
carbon monoxide
- (c)
carbon
- (d)
lime stone
In blast furnace, Fe2O3 is reduced to Fe by CO. Fe2O3 + 3CO ➝ 2Fe + 3CO2 ↑
During extraction of aluminium from bauxite,
- (a)
the concentration of ore is done by gravity separation method
- (b)
molten mixture of aluminium oxide, cryolite or fluorspar is electrolysed
- (c)
impure aluminium is refined by liquation
- (d)
molten aluminium is obtained at cathode while fluorine is liberated at anode.
Purified Al2O3 is mixed with Na3AlF6 and CaF2 to improve conductivity of Al2O3 and to lower the melting point.
\({ Al }_{ 2 }{ O }_{ 3 }\underrightarrow \ { electrolysis } \ { 2Al }^{ 3+ }+{ 3O }^{ 2- }\)
At cathode: Al3++ 3e- ➝ Al
At anode: C + O2- ⟶CO + 2e-
C + 2O2- ➝ CO2 + 4e-
Which process of purification is represented by the following reaction?
\(\underset { Impure }{ Ti } +{ 2I }_{ 2 }\xrightarrow{ { 250 }^{ o }C } { TiI }_{ 4 }\xrightarrow{ 1400^{ o }C } \underset { pure }{ Ti } +{ 2I }_{ 2 }\)
- (a)
Zone refining
- (b)
Monds process
- (c)
Cupellation
- (d)
van Arkel process
Assertion : Nickel is purified by reacting it with CO.
Reason : Impurities present, form a volatile complex.
- (a)
If both assertion and reason are true and reason is the correct explanation of assertion.
- (b)
If both assertion and reason are true but reason is not the correct explanation of assertion
- (c)
If assertion is true but reason is false.
- (d)
If both assertion and reason are false
Ni reacts with CO to form nickel tetracarbonyl
Which of the following pairs of compounds is isoelectronic and isostructural?
- (a)
BeCl2, XeF2
- (b)
Tel2, XeF2
- (c)
lBr2-, XeF2
- (d)
IF3, XeF2
Thus, both IBr2-and XeF2 have linear geometry. So, they are iso-structural and number of valence electrons present in both the species is same, i.e., 22. Thus, they are also isoelectronic.
| S.No. | Compounds | Number of valence electrons | Geometry |
| 1. | BeCl2 | 2+14=16 | Linear |
| 2. | XeF2 | 8+14=22 | Linear |
| 3. | Tel2 | 6+14=20 | Bent or V-shape |
| 4. | lBr2- | 7+14+1=22 | Linear |
| 5. | lF2 | 7+21=28 | T-shape |
Which of the following oxides is anhydride of nitrous acid?
- (a)
N2O3
- (b)
NO2
- (c)
NO
- (d)
N2O4
2HNO2\(\rightarrow \)N2O3 + H2O
Fill in the blanks:
(i) Ca3P2+ 6HCI \(\rightarrow \) 3CaCl2 + .....p .....
(ii) P4 + 3NaOH + 3H2O \(\rightarrow \) ....q.... + 3NaH2PO2
(iii) PH4I + KOH \(\rightarrow \) KI + H2O + ...r... .
p, q and r respectively are
- (a)
(a) PH3, H3PO3, PI3
- (b)
(b) PH3, PH3, PH3
- (c)
(c) PCI3, H3PO4, PH3
- (d)
(d) PCl, PH3, P4O6
Which of the following is a tetrabasic acid?
- (a)
- (b)
- (c)
- (d)
Elements of group-15 form compounds in +5 oxidation state. However, bismuth forms only one well characterised compound in +5 oxidation state. The compound is
- (a)
- (b)
BiF5
- (c)
- (d)
Bi2S5
The stability of +5 oxidation state decreases down the group due to inert pair effect. The only well characterised Bi(V) compound is BiF5 as fluorine being most electronegative element is able to unpair ns electrons.
Hot conc. H2SO4 acts as moderately strong oxidising agent. It oxidises both metals and non- metals. Which of the following elements is oxidised byconc. H2SO4 into two gaseous products?
- (a)
Cu
- (b)
S
- (c)
C
- (d)
Zn
C+2H2SO4(conc.) ⟶ CO2(g)+2SO2(g)+2H2O
In solid state PCl5 is a ___________
- (a)
- (b)
- (c)
- (d)
Complete the following reactions:
(i) SiO2 + 2NaOH ⟶ X + H2O
(ii) SiO2 + 4HF ⟶ Y + 2H2O
(iii) Si + 2CH3CI \(\xrightarrow[570\ K]{Cu\ powder}\) Z
- (a)
X Y Z Na2SiO3 SiF4 (CH3)2SiCl2 - (b)
X Y Z H2SiO3 SiF2 CH3SiCl3 - (c)
X Y Z Na2SiO3 H2SiO3 (CH3)3SiCl - (d)
X Y Z Na2SiO3 H2SiF4 (CH3)2SiCl2
\(SiO_2 + 2NaOH \to \underset{(X)}{Na_2SiO_3} + H_20\)
\(SiO_2 + 4HF \to \underset{(Y)}{SiF_4} + 2H_20\)
\(Si + 2CH_3CI\xrightarrow[570K]{Cu\ powder}\underset{(Z)}{(CH_3)_2SiCl_2}\)
Assertion: CO2 is a gas at room temperature while SiO2 is a crystalline solid.
Reason: SiO2 is a network of silicon and oxygen atoms joined by multiple bonds.
- (a)
- (b)
- (c)
- (d)
SiO2 is a three-dimensional network solid in which each Si atom is covalently bonded in a tetrahedral manner to four O atoms.
For Zn2+, Ni2+, Cu2+ and Cr2+ which of the following statements is correct?
- (a)
- (b)
- (c)
- (d)
Zn2+(3d10) has zero unpaired electron (colourless).
Ni2+(3d8) has 2 unpaired electrons (coloured).
Cu2+(3d9) has 1 unpaired electron (coloured).
Cr2+(3d4) has 4 unpaired electrons (coloured).
A solution of KMnO4 is reduced to various products depending upon its pH. At pH < 7 it is reduced to a colourless solution (A), at pH = 7 it forms a brown precipitate (B) and at pH > 7 it gives a green solution (C). (A), (B) and (C) are:
- (a)
(A) (B) (C) Mn2+ MnO2 MnO42- - (b)
(A) (B) (C) MnO2 Mn2+ MnO42- - (c)
(A) (B) (C) Mn2+ MnO42- MnO2 - (d)
(A) (B) (C) MnO42- Mn2+ MnO2
At pH < 7, in acidic medium
\(MnO_4^-+8H^++5e^-\to\underset{(colourless)}{Mn^{2+}}+4H_2O\)
At pH = 7, in neutral medium
\(MnO_4^-+2H_2O+3e^-\to\underset{(browen\ precipitate)}{MnO_2}+4OH^-\)
At pH > 7, in alkaline medium
\(Mn{ O }_{ 4 }^{ - }+{ e }^{ - }\longrightarrow\underset{(green)} {Mn{ O }_{ 4 }^{ 2- }}\)
The correct configuration off-block elements is:
- (a)
- (b)
- (c)
- (d)
Of the following complex ions which is diamagnetic in nature?
- (a)
[Ni(CN)4]2-
- (b)
[CuCl4]2-
- (c)
[COF6]3-
- (d)
[NiCl4]2-
For [Ni(CN)4]2-
Ni+2 = 3d8 4s0
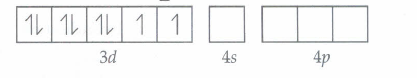
CN- is a strong field ligand thus, it pair up the electrons.
.png)
Thus, [Ni(CN)4]-2 is diamagnetic in nature.
Among [Ni(CO)4], [Ni(CN)4]2-, [NiCl4]2- species, the hybridization states of the Ni atom are, respectively (Atomic number of Ni = 28)
- (a)
sp3, dsp2, dsp2
- (b)
sp3, dsp2, sp3
- (c)
sp3, sp3, dsp2
- (d)
dsp2, sp3, sp3
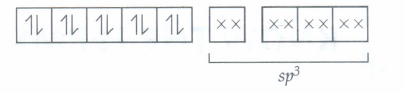
In Ni(CO)4, Nickel is sp3 hybridised because in it oxidation state of Ni is zero.
28Ni = [Ar] 3d6 4s2
(CO is a strong field ligand hence does the pairing of electrons) In [Ni(CN)4]2- nickel is present as Ni+2, so its configuration is
[Ar]3d8 4s0
.png)
CN- is a strong field ligand so pairing occurs. In [NiCl4]2-
Ni+2 = [Ar]3d8 4s0
.png)
Cl- is a weak field ligand hence electrons are not paired.
Ammonia acts as a very good ligand but ammonium ion does not form complexes because
- (a)
- (b)
- (c)
- (d)
Low spin tetrahedral complexes are not formed because
- (a)
- (b)
- (c)
- (d)
Crystal field stabilisation energy for tetrahedral complexes is less than pairing energy hence they do not pair up to form low spin complexes
Which of the following will give enantiomeric pair on reaction with water due to presence of asymmetric carbon atom?
- (a)
\({ C }_{ 2 }{ H }_{ 5 }-\overset { \underset { | }{ { { C }_{ 2 } }{ H }_{ 5 } } }{ \underset { \overset { | }{ { C }_{ 2 }{ H }_{ 5 } } }{ C } } -Br\)
- (b)
\({ C }_{ 2 }{ H }_{ 5 }-\overset { \underset { | }{ { C }_{ 2 }{ H }_{ 5 } } }{ \underset { \overset { | }{ { CH }_{ 3 } } }{ C } } -CI\)
- (c)
\({ C }_{ 2 }{ H }_{ 5 }-\overset { \underset { | }{ H } }{ \underset { \overset { | }{ { CH }_{ 3 } } }{ C } } -I\)
- (d)
\({ C }_{ 2 }{ H }_{ 5 }-\overset { \underset { | }{ { CH }_{ 3 } } }{ \underset { \overset { | }{ { C }_{ 2 }{ H }_{ 5 } } }{ C } } -Br\)
Option (c) has asymmetric carbon atom which is joined to four different groups hence it will give optically active products or a pair of enantiomers.
Match the reactions given in column I with the type of reaction mentioned in column II and mark the appropriate choice.
| Column I | Column II | ||
|---|---|---|---|
| (A) | (i) | \(\beta \)-elimination | |
| (B) | CH3CH2Br \(\xrightarrow{AgOH}\) CH3CH2OH | (ii) | SN1 nucleophilic substitution |
| (C) | CH3CH = CH2 + HBr \(\xrightarrow { peroxide }\) CH3CH2CH2Br | (iii) | SN2 nucleophilic substitution |
| (D) | CH3 - CH2Br + ale. KOH ⟶ CH2 = CH2 | (iv) | Kharasch effect |
- (a)
(A) ⟶ (iv), (B) ⟶ (i), (C) ⟶ (ii), (D) ⟶ (iii)
- (b)
(A) ⟶ (ii), (B) ⟶ (iii), (C) ⟶ (iv), (D) ⟶ (i)
- (c)
(A) ⟶ (i), (B) ⟶ (ii), (C) ⟶ (iv), (D) ⟶ (iii)
- (d)
(A) ⟶ (iii), (B) ⟶ (i), (C) ⟶ (ii), (D) ⟶ (iv)
Assertion: SN1 reactions are generally carried out in polar protic solvents (like water, alcohol, acetic acid, etc.)
Reason : C6H5CH(C6H5)Br is less reactive than C6H5CH(CH3)Br in SN1 reactions.
- (a)
- (b)
- (c)
- (d)
Carbocation obtained from C6H5CH(C6Hs)Br is more stable than C6H5CH(CH3)Br as C6H5CH(C6H5) is stabilised by two phenyl groups and hence it is more reactive in SN1 reaction.
An equimolar quantities of ethanol and propanol is heated with cone. H2SO4, The product formed is/are
- (a)
C2H5OC2H5
- (b)
- (c)
- (d)
C2H5OH + CH3CH2CH2OH \(\\ \overset { cone.H_{ 2 }S_{ 4 } }{ \underset { \Delta }{ \longrightarrow } } \)
C2H5OC2H5+ C3H7OC3H7 + C2H5OC3H7
Assertion: The relative ease of dehydration of alcohols follow the following order:
Tertiary > Secondary > Primary
Reason: Formation of carbocation is the slowest step of the reaction
- (a)
- (b)
- (c)
- (d)
Which of the following reagents would distinguish cis-cyclopenta-1, 2-diol from the trans-isomer?
- (a)
MnO2
- (b)
Aluminium isopropoxide
- (c)
Acetone
- (d)
Ozone
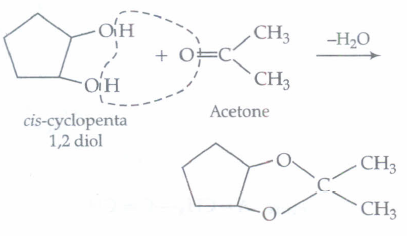
Trans isomer does not react with acetone as removal of H2O molecule is difficult.
Ketones \([R-\underset { \overset { || }{ O } }{ C } -{ R }_{ 1 }],\) where R = R1 = alkyl group, can be obtained in one step by:
- (a)
hydrolysis of esters
- (b)
oxidation of primary alcohol
- (c)
oxidation of tertiary alcohol
- (d)
reaction of acid halide with alcohol
By oxidation of tertiary alcohol with stronger oxidizing agent can yield ketones along with carboxylic acid.
Carboxylic acids have higher boiling points I than aldehydes, ketones and even alcohols of I comparable molecular mass. It is due to their:
- (a)
More extensive association of carboxylic acid via van der Waals force of attraction
- (b)
Formation of carboxylate ion
- (c)
Formation of intramolecular H-bonding
- (d)
Formation of intermolecular H-bonding
Due to formation of intermolecular H-bonding in carboxylic acid, association occurs. Hence boiling point increases and become more than the boiling point of aldehydes, ketones and alcohols of comparable molecular masses.
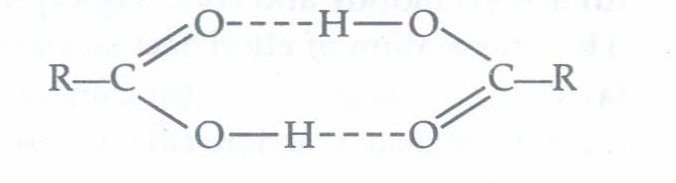
Identify reactant (X) in the given reaction sequence.
\({ CH }_{ 3 }CO{ CH }_{ 3 }+X\longrightarrow \left( { CH }_{ 3 } \right) _{ 3 }C-OKg-CI\overset { { H }_{ 2 }O }{ \longrightarrow \left( { CH }_{ 3 } \right) _{ 3 }C-OH+M{ g }_{ { \diagdown }_{ CI } }^{ { \diagup }^{ OH } } } \)
- (a)
CH3MgCI
- (b)
CH3COCl + Mg
- (c)
MgCI2
- (d)
CH3CH2MgCI
On boiling the egg, what structural changes are taking place in the egg white?
- (a)
- (b)
- (c)
- (d)
During denaturation of proteins, 2° and 3° structures are destroyed but primary structure remains intact.
Regarding cross-linked or network polymers, which of the following statement is incorrect?
- (a)
Examples are bakelite and melamine
- (b)
They are formed from bi- and tri-functional monomers
- (c)
They contain covalent bonds between various linear polymer chains
- (d)
They contain strong covalent bonds in their polymer chains.
Cross-linked or network polymers are usually formed from bi-functional & tri-functional monomers and contains strong covalent bond between various linear polymer chains like Melamine, Bakelite etc.
Which one of the following statements is not true?
- (a)
Ampicillin is a natural antibiotic
- (b)
Aspirin is both analgesic and antipyretic
- (c)
Sulphadiazine is a synthetic antibacterial drug
- (d)
Some disinfectants can be used as antiseptics.
Ampicillin is a modification of penicillin and not a natural antibiotic. These semisynthetic penicillin (SSP) like ampicillin, cloxacillin etc., are produced by chemically combining specific side chains (in place of benzyl side chain of penicillin group) or by incorporating specific precursors in the mould cultures.
Which of the following statements is not correct?
- (a)
- (b)
- (c)
- (d)
Disinfectants are harmful to the living tissues hence they are not applied to the skin.
Which among the given molecules can exhibit tautomerism?
- (a)
III only
- (b)
Both I and III
- (c)
Both I and II
- (d)
Both II and III
α-hydrogen at bridge carbon never participates in tautomerism. Thus only (III)exhibits tautomerism
Which of the following intermediates contains three pairs of electrons in its valence shell?
- (a)
Carbocations
- (b)
Carbanions
- (c)
- (d)
The increasing order of electron donating inductive effect of alkyl groups is
- (a)
- (b)
- (c)
- (d)
The blue compound formed in the positive test for nitrogen with Lassaigne solution of an organic compound is
- (a)
- (b)
- (c)
- (d)
A sample of 0.50 g of an organic compound was treated according to Kjeldahl's method. The ammonia evolved was absorbed in 50 mL of 0.5 M H2SO4. The residual acid required 60 mL of 0.5 M solution of NaOH for neutralisation. What would be the percentage composition of nitrogen in the compound?
- (a)
50
- (b)
- (c)
- (d)
Given:
Mass of compound taken = 0.50 g
Vol. of H2SO4 = 50 mL
Molarity of H2SO4 = 0.5M
Vol.of NaOH required = 60 mL
Molarity of NaOH required = 0.5 M
Method adopted: Kjeldahl's method
Formula used: % of N =\(\cfrac { 1.4\times M\times 2\left[ V-\cfrac { { V }_{ 1 } }{ 2 } \right] }{ m } \)
By substituting the values in the formula, we get,
% of N = \(\cfrac { 1.4\times 0.5\times 2\left( 50-60/2 \right) }{ 0.5 } =56\)
. . % of N in the given compound = 56%







1675494153.jpg)

1675329406.jpg)
1652956171.jpg)

