

NEET 2020 Mock Test 3
Exam Duration: 180 Mins Total Questions : 180
The magnitudes of vectors \(\overrightarrow { A } ,\overrightarrow { B } \) and \(\overrightarrow { C }\) are respectively 12 5 and 13 units and \(\overrightarrow { A } +\overrightarrow { B } \)=\(\overrightarrow { C }\), then the angle between \(\overrightarrow { A }\) and \(\overrightarrow { B }\) is:
- (a)
0
- (b)
\(\pi\)
- (c)
\(\pi\)
- (d)
\(\frac{\pi}{4}\)
If \(\vec{A}\) and \(\vec{B}\) are two vectors, which of the following is not correct?
- (a)
\(\vec { A } +\vec { B } =\vec { B } +\vec { A } \)
- (b)
\(\vec { A }.\vec { B } =\vec { B } .\vec { A } \)
- (c)
\(\vec { A } \times \vec { B } =\vec { B } \times \vec { A } \)
- (d)
\(\vec { A } -\vec { B } =-(\vec { B } -\vec { A } )\)
A car is moving horizontally along a straight line with a uniform velocity of 25 m s -1. A projectile is to be fired from this car in such a way that it will return to it after it has moved 100 m. The speed of the projectile must be:
- (a)
9.8 ms-1
- (b)
19.6 ms-1
- (c)
15.6 ms-1
- (d)
24.6 ms-1
The order of magnitude of the diameter of the earth is (Diameter of the earth is 1.28 x 107m)
- (a)
5
- (b)
6
- (c)
7
- (d)
8
The cell wall is composed of two thin overlapping shells which fit together like a soap case in
- (a)
desmids
- (b)
diatoms
- (c)
dinoflagellates
- (d)
slime moulds.
Which of the following statements regarding Kingdom Animalia is incorrect?
- (a)
It includes heterotrophic, unicellular eukaryotic organisms.
- (b)
The members of this kingdom lack cell walls.
- (c)
The mode of nutrition is holozoic.
- (d)
The sexual reproduction in its members is by copulation of male and female.
Select the pair that consists of viral diseases
- (a)
Mumps and small pox
- (b)
Herpes and influenza
- (c)
Pneumonia and syphilis
- (d)
Both (a) and (b)
The most efficient locomotion in protists is through:-
- (a)
Pseudopodia
- (b)
Flagella
- (c)
Cilia
- (d)
Tentacles
Neurospora, which is popularly known as Drosophila of plant kingdom, belongs to:-
- (a)
Phycomycets
- (b)
Ascomycetes
- (c)
Basidiomycetes
- (d)
Seuteromycetes
Peat moss is
- (a)
Sphagnum
- (b)
Dryopteris
- (c)
Funaria
- (d)
Polytrichum
Which one of the following pairs is wrongly matched?
- (a)
Coliforms-Vinegar
- (b)
Methanogens-Gobar gas
- (c)
Yeast-Ethanol
- (d)
Streptomycetes-Antibiotic
Which one of these animals is not a homeotherm ?
- (a)
Camelus
- (b)
Cheone
- (c)
Macropus
- (d)
Psittacula
Body cavity is the cavity present between body wall and gut wall. In some animals the body cavity is not lined by mesoderm. Such animals are called
- (a)
acoelomate
- (b)
pseudocoelomate
- (c)
coelomate
- (d)
haemocoelomate.
Reticulate venation is a characteristic of dicots. An exception to this generalisation is
- (a)
Ca/ophyllum
- (b)
Ficus
- (c)
Hibiscus
- (d)
Zizyphus.
A small rootless aquatic herb in which a portion of leaf forms a tiny sack or bladder which traps water insects is
- (a)
Dionaea
- (b)
Utricularia
- (c)
Sarracenia
- (d)
Drosera.
Match the columns and choose the correct option
| Column I (Fruit) | Column II (Edible part) |
|---|---|
| a) Walnut | I) Cotyledon |
| b) Cashewnut | II) Seed |
| c) Orange | III) Endocarp |
| d) Strawberry | IV) Thalamus |
- (a)
a-II, b-I, c-III, d-IV
- (b)
a-II, b-III, c-I, d-IV
- (c)
a-I, b-II, c-IV, d-III
- (d)
a-I, b-II, c-III, d-IV
When adventitious roots are shallow surface feeders then they are known as
- (a)
Tuberous root
- (b)
Prop root
- (c)
Fibrous root
- (d)
Conial root
Find the odd one w.r.t stem tendril
- (a)
Grapevines
- (b)
Cucumber
- (c)
Pea
- (d)
Pumpkin
Zygomorphic flower occurs in
- (a)
Pea
- (b)
Gulmohur
- (c)
Cassia
- (d)
All of these
Stomata are distributed more on the lower surface than on the upper surface in
- (a)
equifacial leaf
- (b)
bifacial leaf
- (c)
unifacial leaf
- (d)
both (a) and (b).
In a dorsiventral leaf, what is true regarding the position of xylem?
- (a)
Xylem is towards adaxial epidermis.
- (b)
Xylem is towards abaxial epidermis.
- (c)
Xylem surrounds phloem.
- (d)
Xylem is surrounded by phloem.
Cell wall in dead mechanical tissue show
- (a)
Lignified nature
- (b)
Cutinised nature
- (c)
Pectose deposition
- (d)
Hemicellulose deposition
Heart wood is
- (a)
Situated away form vascular cambium
- (b)
Situated near pith
- (c)
Nonfunctional
- (d)
All of these
Assertion: Tendons attach one bone to another bone.
Reason: Ligaments attach skeletal muscles to bones.
- (a)
If both assertion and reason are true and reason is the correct explanation of assertion.
- (b)
If both assertion and reason are true but reason is not the correct explanation of assertion.
- (c)
If assertion is true but reason is false.
- (d)
If both assertion and reason are false.
Which of the following describes the given graph correctly?
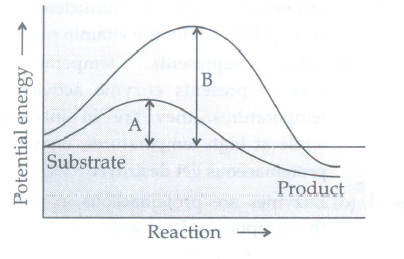
- (a)
Endothermic reaction with energy A in presence of enzyme and B in absence of enzyme.
- (b)
Exothermic reaction with energy A in presence of enzyme and B in absence of enzyme.
- (c)
Endothermic reaction with energy A in absence of enzyme and B in presence of enzyme.
- (d)
Exothermic reaction with energy A in absence of enzyme and B in presence of enzyme.
In meiosis crossing over is initiated at:
- (a)
Diplotene
- (b)
Pachytene
- (c)
Leptotene
- (d)
Zygotene
At what phase of meiosis there are two cells, each with separated sister chromatids that have been moved to opposite spindle poles?
- (a)
Anaphase II
- (b)
Anaphase I
- (c)
Telophase II
- (d)
Telophase I
In meiosis, how many cycles of chrmosome division occurs?
- (a)
One
- (b)
Four
- (c)
Two
- (d)
Three
During cell division, spindle fibers attach to which part of chromosome ;
- (a)
Primary constriction
- (b)
Sec, constriction
- (c)
Chromomere
- (d)
Chromatid
Ascent of sap is best explained by
- (a)
- (b)
- (c)
- (d)
Water moves up against gravity and even for a tree of 20 m height, the tip receives water within two hours. The most important physiological phenomenon which is responsible for the upward movement of water is _______________.
- (a)
guttation
- (b)
evaporation
- (c)
transpiration
- (d)
Specialised epidermal cells surrounding the guard cells are called
- (a)
- (b)
- (c)
Bulliform cells
- (d)
Lenticels
Select the correctly matched pair
- (a)
- (b)
- (c)
- (d)
Which of the foolowing set contains macro nutrients?
- (a)
- (b)
- (c)
- (d)
Read the given statements and select the correct option.
Statement 1: Carboxylation is the most crucial step of Calvin cycle where CO2 is utilised for the carboxylation of RuBP.
Statement 2: Carboxylation is catalysed by the enzyme RuBisCO which results in the formation of two molecules of 3PGA.
- (a)
- (b)
- (c)
- (d)
What is true about the end products of glycolysis?
- (a)
- (b)
- (c)
- (d)
In the following question, a statement of assertion is followed by a statement of reason. Mark the correct choice as :
Assertion: Fermentation is the incomplete oxidation of glucose into lactic acid or ethanol.
Reason: It takes place under anaerobic conditions in prokaryotes only.
- (a)
- (b)
- (c)
- (d)
Pyuvate dehydrogenase complex is used ion converting-
- (a)
Pyurvate to glucose
- (b)
- (c)
- (d)
Consider the following statements each with one or two blanks.
(i) Left lung has___(1)___lobes and right lung has___(2)____lobes.
(ii) Prawn respires with___(3)_____and insects with____(4)____.
(iii) Amount of air inhaled and exhaled with maximum effort is referred to as the___(5)____of the lungs.
Fill up the above blanks by selecting the correct option.
- (a)
- (b)
- (c)
- (d)
People living at sea level have around 5 million RBC per cubic millimetre of their blood whereas those living at an altitude of 5400 metres have around 8 million. This is because at high altitude
- (a)
- (b)
- (c)
- (d)
Assertion: Vocal cords consist of three pairs of mucous membrane that extend into the lumen of the larynx.
Reason: Sound is produced by only two pairs of cords.
- (a)
- (b)
- (c)
- (d)
People who have migrated from the planes to an area adjoining Rohtang Pass about six months back.
- (a)
Have more RBCs and their haemoglobin has a lower binding affinity to oxygen
- (b)
Are not physically fit to play games like football
- (c)
Suffer from altitude sickness with symptoms like nausea, fatigue etc.
- (d)
Have the usual RBCcount but their haemoglobin has very high binding affinity to oxygen
Excessively high heart rate (> 180) can reduce cardiac output because
- (a)
- (b)
- (c)
- (d)
Which one of the following statements in more dilute urine which one of the following statements in regard to the excretion by the human kidneys is correct?
- (a)
Nearly 90% of the glomerular filtrate is reabsorbed by the renal tubules
- (b)
Ascending limb of the loop of Henle is impermeable to electrolytes.
- (c)
Descending limb of loop of Henle is impermeable to water
- (d)
Distal convoluted tubule is incapable of reabsorbing HCO3
A large quantity of fluid is filtered everyday by nephrons in the kidneys but only about 1% of it is excreted as urine. The remaining 99% of the filtrate
- (a)
- (b)
- (c)
- (d)
Human beings are
- (a)
Uricotelic
- (b)
Ureotelic
- (c)
Ammonotelic
- (d)
What is the osmolarity of the filtrate at the hairpin bend of loop of Henle?
- (a)
- (b)
- (c)
- (d)
Which of the following is a source of energy for muscle contraction?
- (a)
Actin
- (b)
ATP
- (c)
Myosin
- (d)
Actomyosin
The joint in which one of the two bones is fixed in its place and bears a peg like process over which the other bone rotates is called
- (a)
- (b)
saddle joint
- (c)
- (d)
During walking talus passes/transmits about the half of the weight of body to
- (a)
cuneiform
- (b)
Cubold
- (c)
Calcaneum
- (d)
Navicular
In a normal pregnant woman, the amount of total gonadotropin activity was assessed. The result expected was :
- (a)
High level of circulating FSH and LH in the uterus to stimulate implantation of the embryo
- (b)
High level of circulating HCG to stimulate endometrial thickening
- (c)
High level of FSH and LH in uterus to stimulate endometrial thickening
- (d)
High level of circulating HCG to stimulate oestrogen and progesterone synthesis
Endocrine glands have_____to carry their secretions to the specific organ.
- (a)
capillaries
- (b)
tubules
- (c)
- (d)
ducts
Which of the following hormones is necessary for the development of secondary sexual characters in human beings?
- (a)
Estrogen
- (b)
FSH
- (c)
Testosterone
- (d)
Which of the following organs in mammals does not consist of a central 'medullary' region surrounded by a cortical region?
- (a)
Ovary
- (b)
Adrenal
- (c)
Liver
- (d)
Kidney
Boron has two stable isotopes, 10B (19%) and 11B (81%). Average atomic weight for boron in the periodic table is:
- (a)
10.8
- (b)
10.2
- (c)
11.2
- (d)
10.0
The frequency of radiation absorbed or emitted when transition occurs between two stationary states with energies E1 (lower) and E2 (higher) is given by
- (a)
v=\(\frac { { E }_{ 1 }+{ E }_{ 2 } }{ h } \)
- (b)
v=\(\frac { { E }_{ 1 }-{ E }_{ 2 } }{ h } \)
- (c)
v=\(\frac { { E }_{ 1 }\times { E }_{ 2 } }{ h } \)
- (d)
v=\(\frac { { E }_{ 2 }-{ E }_{ 1 } }{ h } \)
The energy of the electron in a hydrogen atom has a negative sign for all possible orbits because:
- (a)
when the electron is attracted by the nucleus and is present in orbit n, the energy is emitted and its energy is lowered.
- (b)
when the electron is attracted by the nucleus and is present in orbit n, the energy is absorbed and its energy is increased.
- (c)
when the electron is repelled by the nucleus, the energy is released and its energy is lowered.
- (d)
none of these.
In the case of alkali metals, the covalent character decreases in the order :
- (a)
MCl > MI > MBr > MF
- (b)
MF > MCl > MBr > MI
- (c)
MF > MCl > MI > MBr
- (d)
MI > MBr > MCl > MF
According to molecular orbital theory which of the following lists rank the nitrogen species in terms of increasing bond order?
- (a)
N2- < N2 < N22-
- (b)
N22- < N2- < N2
- (c)
N2 < N22- < N2-
- (d)
N2- < N22- < N2
Representing P, V and T as pressure, volume and temperature respectively, which of the following is the correct representation of Boyle's law?
- (a)
\(V\propto \frac { 1 }{ T } \) (P constant)
- (b)
\(V\propto \frac { 1 }{ P } \) (T constant)
- (c)
PV=RT
- (d)
PV=nRT
If ∆H is the change in enthalpy and ∆E, the change in internal energy accompanying a gaseous reaction?
- (a)
∆H is always greater than ∆E
- (b)
∆H < ∆E only if the number of moles of products is greater than the number of moles of the reactants
- (c)
∆H is always less than ∆E
- (d)
∆H < ∆E only if the number of moles of products is less than the number of moles of the reactants
A system changes from state X to Y with a change in internal energy measuring to 25 kJ mol-1, by a reversible path and returns from Y to X by an irreversible path. What will be the net change in internal energy?
- (a)
25 kJ
- (b)
> 25 kJ
- (c)
< 25 kJ
- (d)
zero
Bond dissociation enthalpies of H2(g) and N2(g) are 436.0 kJ mol-1 and 941.8 kJ mol-1, respectively, and enthalpy of formation of NH3(g) is -46 kJ mol-1 . What are the enthalpy of atomisation of NH3(g) and the average bond enthalpy of N - H bond respectively (in kJ mol-1)?
- (a)
1170.9,390.3
- (b)
117,300
- (c)
300,200
- (d)
2000,1975
In which of the following, the solubility of AgCl will be minimum?
- (a)
0.1M NaNO3
- (b)
Water
- (c)
0.1M NaCl
- (d)
0.1M NaBr
The yield of NH3 in the reaction N2 + 3H2 \(\rightleftharpoons \) 2NH3; \(\Delta \)H = -22.08 kcal mol'-1 is affected by
- (a)
change in pressure and temperature
- (b)
change in temperature and concentration of N2
- (c)
change in pressure and concentration of N2
- (d)
change in pressure, temperature and concentration of N2.
Acidic character of BF3 can be explained on the basis of which of the following concepts?
- (a)
Arrhenius concept
- (b)
Bronsted-Lowry concept
- (c)
Lewis concept
- (d)
Bronsted-Lowry as well as Lewis concept
Which of the following is not a property of hydrogen?
- (a)
It is a colourless, odourless gas.
- (b)
It is highly combustible.
- (c)
It is highly poisonous gas.
- (d)
It is lighter than air.
Magnesium reacts with an element (X) to form an ionic compound. If the ground state electronic configuration of (X) is 1s2 2s2 2p3, the simplest formula for this compound is:
- (a)
Mg2X
- (b)
MgX2
- (c)
Mg2X3
- (d)
Mg3X2
Mark the incorrect statement from the following.
- (a)
Benzene has a planar structure
- (b)
Benzene is an unsaturated hydrocarbon and shows addition reactions like alkenes
- (c)
In benzene carbon uses two p-orbitals for hybridisation.
- (d)
Aromatic hydrocarbons contain high percentage of carbon hence burn with sooty flame.
Dimensional formula of capacitance is :
- (a)
[M-1L-2T4A2]
- (b)
[ML2T4A2]
- (c)
[MLT-4A2]
- (d)
[M-1L-2T-4A-2]
The length and breadth of a rectangle are (5.7 ± 0.1) cm and (3.4 ± 0.2) cm. The area of rectangle with error limits is approximately:
- (a)
(19.4 ± 1)cm2
- (b)
(19.4 ± 2) cm2
- (c)
(19.4±2.5)cm2
- (d)
(19.4±1.5)cm2
A body of mass 2 kg moving on a horizontal surface with an initial velocity of 4 m/sec comes to rest after 2 sec. If one wants to keep this body moving on the same surface with a velocity of 4 m/sec, the force required is:
- (a)
8 N
- (b)
Zero
- (c)
4 N
- (d)
2 N
A body of mass 16 kg accelerates down a smooth inclined plane with a force of 8 N. What is the minimum force required to make it move upwards with the same acceleration?
- (a)
4 N
- (b)
8 N
- (c)
12 N
- (d)
16 N
A Force F1 of 500N is required to push a car of mass 1000 Kg slowly ar constant speed on a leveled road. If a force F2 of 1000N is applied then the acceleration of the car will be
- (a)
zero
- (b)
1.5 ms-2
- (c)
1 ms-2
- (d)
0.5 ms-2
A particle is moving along a circular path of radius 5 m with a uniform speed 5ms -1. What will be the average acceleration when the particle completes half revolution?
- (a)
Zero
- (b)
10 ms-1
- (c)
10 πms-2
- (d)
\(\frac{10}{\pi}\) ms-2
The maximum safe speed of a vehicle over a curved road of radius 150 m is 10 ms -1. If the width of road is 7.5 m, the height of the outer edge is:
- (a)
0.25 m
- (b)
0.50 m
- (c)
0.35 m
- (d)
0.60 m
A particle is projected with a speed v at 45° with the horizontal. The magnitude of angular momentum of the projectile about the point of projection when the particle is at its maximum height his:
- (a)
Zero
- (b)
\(\frac { m\upsilon { h }^{ 2 } }{ \sqrt { 2 } } \)
- (c)
\(\frac { m\upsilon ^{ 2 }h }{ 2 } \)
- (d)
\(\frac { m\upsilon { h }^{ 3 } }{ \sqrt { 2 } } \)
- (e)
\(\frac { m\upsilon { h } }{ \sqrt { 2 } } \)
Two blocks of masses 5 kg and 2 kg are placed on frictionless surface and connected by a spring. An external kick gives a velocity of 14 m/sec to the heavier block in the direction of lighter one. The velocity gained by the centre of mass is:
- (a)
14ms-1
- (b)
7ms-1
- (c)
12ms-1
- (d)
10ms-1
An initial momentum is imparted to a homogeneous cylinder, as a result of which it begins to roll without slipping up an inclined plane at a speed ofvo = 4 m s-1 . The plane makes an angle \(\theta \) = 30° with the horizontal. What height II will the cylinder rise to? ( Take g = 10m s-2)
- (a)
0.8 m
- (b)
1.2 m
- (c)
1.0 m
- (d)
1.6 m
The moment of inertia of a thin circular disc about an axis passing through its centre and perpendicular to its plane is I. Then, the moment of inertia of the disc about an axis parallel to its diameter and touching the edge of the rim is:
- (a)
I
- (b)
2I
- (c)
\(\frac { 3 }{ 2 } I\)
- (d)
\(\frac {5 }{ 2 } I\)
The ratio of the radii of gyration of a circular disc about a tangential axis in the plane of the disc and of a circular ring of the same radius about a tangential axis in the plan of the ring is:
- (a)
\(\sqrt { 3 } :\sqrt { 5 } \)
- (b)
\(\sqrt { 12 } :\sqrt { 3 } \)
- (c)
\( { 1 } :\sqrt { 3 } \)
- (d)
\(\sqrt {5 } :\sqrt { 5} \)
An iron block of sides 50 cm x 8 cm x 15 cm has to be pushed along the floor. The force required will be minimum when the surface in contact with ground is
- (a)
8 cm x 15 cm surface
- (b)
50 cm x 15 cm surface
- (c)
8 cm x 50 cm surface
- (d)
force is same for all surfaces
block of mass 4 kg is placed on a rough horizontal plane. A time dependent force F=kf2 acts on the block, where k = 2 N/s2. Force of friction between block and the plane at t = 2 see is:
- (a)
8 N
- (b)
4 N
- (c)
2 N
- (d)
1 N
If the ball is thrown towards the surface of the earth:
- (a)
the earth remains stationary while the ball moves downwards
- (b)
the ball remains stationary while the earth moves upwards
- (c)
the ball and the earth move towards each other
- (d)
the ball and the earth move away from each other
A particle of mass M, starting from rest, undergoes uniform acceleration. If the speed acquired in time T is V, the power delivered to the particle is:
- (a)
\(\cfrac { { MV }^{ 2 } }{ T } \)
- (b)
\(\cfrac { 1 }{ 2 } \cfrac { MV^{ 2 } }{ { T }^{ 2 } } \)
- (c)
\(\cfrac { { MV }^{ 2 } }{ { T }^{ 2 } } \)
- (d)
\(\cfrac { 1 }{ 2 } \cfrac { MV^{ 2 } }{ { T }^{ } } \)
Consider a system of two particles having masses m1 and m2. If the particle of mass m1 is pushed towards the centre of mass of particles through a distance d, by what distance would the particle of mass m2 move so as to keep the centre of mass of particles at the original position?
- (a)
(m2/m1)d
- (b)
\(\frac {m_1}{m_1 + m_2}d\)
- (c)
(m1/m2)d
- (d)
d
LANDSAT series of satellite move in near polar orbits at an altitude of
- (a)
3600 km
- (b)
3000 km
- (c)
918 km
- (d)
512 km
The mass of the earth is 81 times that of the moon and the radius ofthe earth is 3.5 times that of the moon. The ratio of the escape velocity on the surface of the earth to that on the surface of the moon will be:
- (a)
- (b)
- (c)
- (d)
The earth's radius is R and acceleration due to gravity at its surface is g. If a body of mass m is sent to a height h = \(\frac { R }{ 5 } \) from the earth's surface, the potential energy increases by:
- (a)
mgh
- (b)
\(\frac { 4 }{ 5 } \) mgh
- (c)
\(\frac {5 }{ 6 } \) mgh
- (d)
\(\frac { 6 }{7 } \) mgh
A rubber cord catapult has cross-sectional area 25 nun 2 and initial length of rubber cord is 10 cm. It is stretched to 5 cm and then released to project a missile of mass 5 gm. Taking Yrubber = 5 x 108 N/m2 velocity of projected missile is :
- (a)
20 m/s
- (b)
100 m/s
- (c)
250 m/s
- (d)
200 m/s
The amount of radiation emitted by a perfectly black body is proportional to
- (a)
Temperature on ideal gas scale
- (b)
Fourth root of temperature on ideal gas scale
- (c)
Fourth power of temperature on ideal gas scale
- (d)
Source of temperature on ideal gas scale
In case of a hollow body, if ρB and ρs represent the densities of body and substance respectively, then:
- (a)
- (b)
- (c)
- (d)
If two liquids of same volume but different densities p1 and p2 are mixed, then density of mixture is given by:
- (a)
\(p={p_1p_2\over2}\)
- (b)
\(p={p_1+p_2\over2p_1p_2}\)
- (c)
\(p={2p_1p_2\over p_1+p_2}\)
- (d)
\(p={p_1p_2\over p_1+p_2}\)
A good lubricant should have:
- (a)
- (b)
- (c)
- (d)
high density
In a thermodynamic process, pressure of a fixed mass of a gas is changed in such a manner that the gas releases 20 J of heat and 8 J of work is done on the gas. If initial internal energy of the gas was 30 J, what will be the final internal energy?
- (a)
- (b)
- (c)
- (d)
Which of the following process is correct for the given P - V diagram.
- (a)
Adiabatic process
- (b)
Isothermal process
- (c)
Isobaric process
- (d)
Isochoric process
What is the most likely value for Cr (molar heat capacity at constant temperature)?
- (a)
0
- (b)
- (c)
- (d)
The phase difference between two waves, represented by: \({ y }_{ 1 }={ 10 }^{ -6 }\sin { \left[ 100t+(x/50)+0.5 \right] } m\quad { y }_{ 2 }={ 10 }^{ -6 }\cos { \left[ 100t+(x/50) \right] } m\). where x is expressed in metres and t is expressed in seconds, is approximately:
- (a)
1.07 rad
- (b)
2.07 rad
- (c)
0.5 rad
- (d)
1.5 rad
41 tuning forks are arranged such that every fork gives 5 beats with the next. The last fork has a frequency that is double of the first. The frequency of the first fork is:
- (a)
- (b)
- (c)
- (d)
In ginger vegetative propagation occurs through
- (a)
Runners
- (b)
Rhizome
- (c)
Offsets
- (d)
Bulbils
Which one of the following pollination is autogamous?
- (a)
Cleistogamy
- (b)
Suspensor
- (c)
Egg
- (d)
Synergid
In female gametophyte which cell is mother cell of endosperm?
- (a)
Synergid
- (b)
Antipodal
- (c)
Central cell
- (d)
Egg
Fertilization in humans is practically feasible only if
- (a)
The ovum and sperms are transported simultaneously to ampullary - isthmic junction of the fallopian tube
- (b)
The ovum and sperms are transported simultaneously to ampullary - isthimic junction of the cervix
- (c)
The sperms are transported into cervix within 48 hours of release to ovum in uterus
- (d)
The sperms are transported into vagina just after the release of ovum in fallopian tube
Which one of the following groups includes sexually transmitted infections caused by bacteria only?
- (a)
- (b)
- (c)
- (d)
Which of the following statements is correct?
- (a)
- (b)
- (c)
- (d)
A plant with genotype AABBCC is selfed F2 phenotypic ratio would be :
- (a)
9: 3: 3: 1
- (b)
27: 9: 9: 9: 3: 3: 3
- (c)
1: 1
- (d)
3: 1
When a heterozygous tall pea plant of F1 generation upon self fertilization produces tall and dwarf phenotypes it proves the principle of
- (a)
Dominance
- (b)
Segregation
- (c)
Independant assortement
- (d)
Complete linkage is found in
- (a)
Birds
- (b)
Snakes
- (c)
- (d)
A nutritionally wild type organism, which does not require any additional growth supplement is known as:
- (a)
prototroph
- (b)
phenotype
- (c)
Holotype
- (d)
Auxotroph
Which one of the following conditions correctly describes the manner of determining the sex in the given example?
- (a)
- (b)
- (c)
- (d)
When the codon of mRNA is 5'-GUC-3' then the anticodon on tRNA will be
- (a)
- (b)
- (c)
- (d)
Severo Ochoa enzyme is
- (a)
- (b)
- (c)
- (d)
During protein synthesis, amino acid gets attached to tRNA with the help of
- (a)
mRNA
- (b)
- (c)
Ribosome
- (d)
rRNA
The effects of genetic drift are more marked in
- (a)
- (b)
- (c)
- (d)
An auto-immune disease is
- (a)
SCID
- (b)
rheumatoid arthritis
- (c)
- (d)
Which of the following statements is correct regarding nectarless cotton varieties?
- (a)
- (b)
- (c)
- (d)
Tropical cane grown in south india is
- (a)
- (b)
- (c)
- (d)
Frieswal is Crossbreed of
- (a)
- (b)
- (c)
- (d)
Which of the following is not a component of downstream processing?
- (a)
Separation
- (b)
Purification
- (c)
Preservation
- (d)
Expression
Stirred-tank bioreactors have been designed for:
- (a)
Purification of product
- (b)
Addition of preservatives to the product
- (c)
Availability of oxygen throughout the process
- (d)
Ensuring anaerobic conditions in the culture vessel
In addition to Taq polymerase enzyme which other thermostable DNA polymerases have been isolated to be used in polymerase chain Reaction (PCR)?
- (a)
- (b)
- (c)
- (d)
A piece of nucleic acid using to find out a gene, by forming hybrid with it, is called as :
- (a)
- (b)
DNA probe
- (c)
- (d)
Which variety of rice was patented by a U.S. company even though the highest number of varieties of this rice are found in India?
- (a)
Sharbati Sonora
- (b)
- (c)
- (d)
Natality refers to:
- (a)
Number of individuals leaving the habitat
- (b)
Birth rate
- (c)
Death rate
- (d)
Number of individuals entering a habitat
Gause's principle of competitive exclusion states that:
- (a)
More aboundant species will exclude the less abundant species through competition
- (b)
Competition for the same resources excludes species having food preferences
- (c)
No two species can occupy the same niche indefinitely for the same limiting resources
- (d)
Larger organisms exclude smaller ones through competition
Which of the following equations correctly represents Verhulst-Pearl logistic growth?
- (a)
dN/dt = rN\(\left( \frac { K-N }{ K } \right) \)
- (b)
dN/dt = \(\frac{rN}{K}\)
- (c)
dN/dt = \(\frac { N\left( K-N \right) }{ K } \)
- (d)
dN/dt = \(\frac { r\left( K-N \right) }{ K } \)
The plant - animal interactions often involve co-evolution of the mutualists so that
- (a)
- (b)
- (c)
the animal utilises plant not only for oviposition but also to pollinate the plant
- (d)
Where would the greatest number of endemic species occur and why?
- (a)
- (b)
- (c)
- (d)
Phosphate pollution is brought about by
- (a)
- (b)
- (c)
- (d)
Which of the following are the correct approaches to reduce global warming?
(i) Use of fossil fuels
(ii) Improving efficiency of energy usage
(iii) Afforestation
(iv) Increasing growth of human population
- (a)
- (b)
- (c)
- (d)
How many chloride ions are surrounding sodium ion in sodium chloride crystal?
- (a)
4
- (b)
8
- (c)
6
- (d)
12
Which of the following will have metal deficiency defect?
- (a)
NaCl
- (b)
FeO
- (c)
KCI
- (d)
ZnO
Experimentally it was found that a metal oxide has formula M0.98O. Metal M, is present as M2+ and M3+ in its oxide. Fraction of the metal which exists as M3+ would be:
- (a)
5.08%
- (b)
7.01%
- (c)
4.08%
- (d)
6.05%
Assertion: Quartz glass is crystalline solid and quartz is an amorphous solid.
Reason: Quartz glass has long range order.
- (a)
If both assertion and reason are true and reason is the correct explanation of assertion.
- (b)
If both assertion and reason are true but reason is not the correct explanation of assertion
- (c)
If assertion is true but reason is false.
- (d)
If both assertion and reason are false
What will be the molality of a solution of glucose in water which is 10% w/W?
- (a)
0.01 m
- (b)
0.617 m
- (c)
0.668 m
- (d)
1.623 m
The molality of 648 g of pure water is:
- (a)
36 m
- (b)
55.5 m
- (c)
3.6 m
- (d)
5.55 m
The standard EMF of a galvanic cell involving cell reaction with n =2 is found to be 0.295 V at 250C. The equilibrium constant of the reaction would be: (Given F = 96500 C mol-1, R = 8.34 JK-1 mol-1)
- (a)
2.0 x 1011
- (b)
4.0 x 1012
- (c)
1.0 x 102
- (d)
1.0 X 1010
Standard reduction potentials at 250C of Li+/Li, Ba2+/Ba, Na+/Na and Mg2+/Mg are -3.05, -2.90, -2.71 and -2.37 V respectively. Which one of the following is the strongest oxidizing agent?
- (a)
Mg2+
- (b)
Ba2+
- (c)
Na+
- (d)
Li+
Molar conductivity of 0.025 mol L-1 methanoic acid is 46.1 S cm2 mol-1, the degree of dissociation and dissociation constant will be
(Given : \({ \lambda }_{ { H }^{ + } }^{ 0 }\)= 349.6 S cm2 mol-1 and \({ \lambda }_{ { H }COO }^{ 0 }\) cm2 mol-1)
- (a)
11.4%, 3.67 x 10-4 mol L-1
- (b)
22.8%, 1.83 x 10-4 mol L-1
- (c)
52.2%, 4.25 x 10-4 mol L-1
- (d)
1.14%,3. 67 x 10-6 mol L-1
An electric current is passed through silver nitrate solution using silver electrodes. 15.25 g of silver was found to be deposited on cathode. What will be the weight of copper deposited on cathode if same amount of electricity is passed through copper sulphate solution using copper electrodes?
- (a)
4.49 g
- (b)
6.4 g
- (c)
12.5 g
- (d)
3.2 g
Assertion: The rate of a reaction sometimes does not depend on concentrations.
Reason: Lower the activation energy, faster is the reaction.
- (a)
If both assertion and reason are true and reason is the correct explanation of assertion
- (b)
If both assertion and reason are true but reason is not the correct explanation of assertion
- (c)
If assertion is true but reason is false
- (d)
If both assertion and reason are false
Which of the following process is not responsible for the presence of electric charge on the sol particles?
- (a)
Electron capture by sol particles
- (b)
Adsorption of ionic species from solution
- (c)
Formation of Helmholtz electrical double layer
- (d)
Absorption of ionic species from solution
For which of the following ores froth floatation method is used for concentration?
- (a)
Haematite
- (b)
Zinc blende
- (c)
Magnetite
- (d)
Carnallite
Carnallite on electrolysis gives
- (a)
Mg and Cl2
- (b)
Ca and Cl2
- (c)
K and Cl2
- (d)
AI and Cl2
Match the inter-halogen compounds of column I with the geometry in Column II and assign the correct code.
| Column I | Column II |
| A.XX' | (i) T-shade |
| B.XX'3 | (ii) Pentagonal bipyramidal |
| C.XX'5 | (iii) Linear |
| D.XX'7 | (iv) Square pyramidal |
| (v)Tetrahedral |
code A B C D
- (a)
(iii) (iv) (i) (ii)
- (b)
(iii) (i) (iv) (ii)
- (c)
(v) (iv) (iii) (ii)
- (d)
(iv) (iii) (ii) (i)
When three parts of conc. HCI and one part of conc. HNO3 is mixed, a compound 'X' is formed. The correct option related to 'X' is
- (a)
- (b)
- (c)
- (d)
Which is not the use of orthoboric acid?
- (a)
- (b)
- (c)
- (d)
Which of the following d-block element has halffilled penultimate as well as valence subshell?
- (a)
Cu
- (b)
Au
- (c)
Ag
- (d)
Cr
n-propyl alcohol and isopropyl alcohol can be chemically distinguished by which reagent:
- (a)
PCl5
- (b)
reduction
- (c)
oxidation with potassium dichromate
- (d)
ozonolysis.
Which of the following compounds does not react with NaOH?
- (a)
- (b)
CH3CONH2
- (c)
- (d)
Assertion: Addition reaction of water to but-l-ene in acidic medium yields butan-1-ol.
Reason: Addition of water in acidic medium proceeds through the formation of primary carbocation.
- (a)
- (b)
- (c)
- (d)
Which of the following structures is not correctly matched?
- (a)
\(\alpha -Mwthoxypropionaldehyde-{ H }_{ 3 }C-\overset { \underset { | }{ { H }_{ 3 }CO } }{ CH } -\overset { \underset { || }{ O } }{ C } -H\)
- (b)
\(3-Hydroxbutanal-{ CH }_{ 3 }-\overset { \underset { | }{ OH } }{ CH } -{ CH }_{ 2 }CHO\)
- (c)
\(4-Ohopentanal-{ CH }_{ 3 }{ CH }_{ 2 }{ CH }_{ 2 }-\overset { \underset { || }{ O } }{ C } -CHO\)
- (d)
\(Di-sec.butlyketone-{ CH }_{ 3 }{ CH }_{ 2 }-\overset { \underset { | }{ { CH }_{ 3 } } }{ CH } -\overset { \underset { || }{ O } }{ C } -\overset { \underset { | }{ { CH }_{ 3 } } }{ CH } -{ CH }_{ 2 }{ CH }_{ 3 }\)
Which of the following statements is not correct?
- (a)
- (b)
- (c)
- (d)
Assertion: The monomer of neoprene is 1, 3-butadiene.
Reason: Neoprene is highly inflammable.
- (a)
- (b)
- (c)
- (d)
What is tincture of iodine?
- (a)
- (b)
- (c)
- (d)
A current I flows in a rectangularly shaped wire whose center lies at (xo, 0, 0) and whose vertices are located at the points A(xo + d, -a, -b), B(xo - d, a, -b), C(xo - d, a, b), and D(xo + d, -a, b) respectively. Assume that a, b, d < < xo. Find the magnitude of magnetic dipole moment vector of the rectangular wire frame. (Given: b = 10 m, d = 4 m, a = 3 m, I = 0.01 A)
- (a)
2 J T-1
- (b)
4 J T-1
- (c)
3 J T-1
- (d)
9 J T-1
A circular coil of 300 turns and diameter 14 cm carries a current of 15 A. The magnitude of magnetic moment associated with the loop is
- (a)
51.7 JT-1
- (b)
69.2 JT-1
- (c)
38.6 JT-1
- (d)
19.5 JT-1
An infinitely long thin wire carrying a uniform linear static charge density A is placed along the z-axis. The wire is set into motion along its length with a uniform velocity v = \(v \vec{k}z\). Find the poynting vector.
- (a)
\(\frac{-\lambda^{2} v }{4\pi^{2} \epsilon_{0}a^{2}}\hat{k}\)
- (b)
\(\frac{\lambda^{2} v }{4\pi^{2} \epsilon_{0}a^{2}}\hat{i}\)
- (c)
\(\frac{\lambda^{2} v }{4\pi^{2} \epsilon_{0}a^{2}}\hat{k}\)
- (d)
\(\frac{-\lambda^{2} v }{4\pi^{2} \epsilon_{0}a^{2}}\hat{i}\)
In the propagation of electromagnetic waves the angle between the direction of propagation and plane of polarisation is
- (a)
0°
- (b)
45°
- (c)
90°
- (d)
180°
Ratio of longest wave lengths corresponding to Lyman and Balmer series in hydrogen spectrum is:
- (a)
5/27
- (b)
3/23
- (c)
7/29
- (d)
9/3
Radioactive material 'N has decay constant '8\(\lambda \)' and material 'B' has decay constant '\(\lambda \)'. Initially they have same number of nuclei. After what time, the ratio of number of nuclei of material 'B' to that 'N will be 1/e?
- (a)
1/7\(\lambda \)
- (b)
1/8\(\lambda \)
- (c)
1/9\(\lambda \)
- (d)
1/\(\lambda \)
Experimental evidence for the existence of the atomic nucleus comes from:
- (a)
Millikan's oil drop experiment
- (b)
Atomic emission spectroscopy
- (c)
The magnetic bending of cathode rays
- (d)
Alpha scattering by a thin metal foil
For a common emitter circuit if IC/IE = 0.98 then current gain for common emitter circuit will be.
- (a)
49
- (b)
98
- (c)
4.9
- (d)
25.5
Correct increasing order of acidity is given as:
- (a)
H2O, C2H2, H2CO3, phenol
- (b)
C2H2, H2O, H2CO3, phenol
- (c)
Phenol, C2H2, H2CO3, H2O
- (d)
C2H2, H2O, phenol and H2CO3







1675494153.jpg)

1675329406.jpg)
1652956171.jpg)

